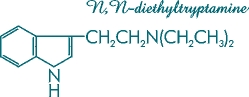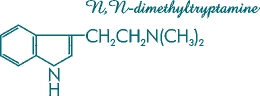Dr. Albert Hofmann, the brilliant Swiss chemist, philosopher, author, and retired Director of the Pharmaceutical-Chemical Research Laboratories of Sandoz Ltd., Basel, is best known for fathering his "problem child," LSD, on April 19, 1943. Thirteen years later, a brief article in a local paper caught his eye. The article stated that a researcher from America had traveled to southern Mexico and participated in a native ritual where mushrooms were consumed that produced strange visions. Already intimately acquainted with the molecular structures of the known psychedelics, Dr. Hofmann was curious about the chemical constituency of the mushrooms. The researcher was, of course, R. Gordon Wasson, but his name was not mentioned in the article and Dr. Hofmann would remain interested in the mysterious mushrooms.
A year later Dr. Hofmann was contacted by Professor Roger Heim, esteemed French mycologist and Director of the Museum National d'Histoire Naturelle in Paris, who requested his assistance in carrying out the chemical investigations of the sacred Mexican mushrooms. Roger Heim had accompanied R. Gordon Wasson on his 1956 expedition to Huautla de Jimenez and identified and named several of the species used by the Mazatecs in their divinatory and curing rites. His beautiful watercolor renderings of sacred Psilocybes accompanied Wasson's Life article in 1957.
Dr. Hofmann enthusiastically accepted Heim's invitation and quickly began the arduous task of isolating the active principles. Also at about this time, unknown to Dr. Hofmann, James Moore -- who had claimed to be a chemist from the University of Delaware -- was working to isolate the mushrooms' actives. His intentions, however, were different from those of Dr. Hofmann's.
In 1956, Moore, who was in reality a CIA operative specializing in the synthesis of psychoactive and chemical weapons for the CIA, offered R. Gordon Wasson a $2,000 grant from the agency's front group, The Geschikter Foundation, and invited himself along on Wasson's next expedition. Wasson, like Dr. Hofmann, had no idea as to Moore's true identity. Moore was hoping to obtain samples of the mushrooms, isolate their active principles and provide the CIA with some new "mind-control" toys.
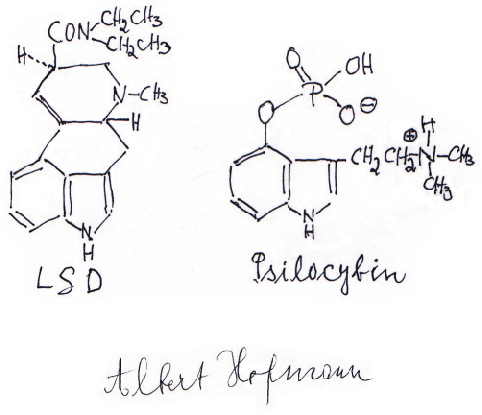
Above: Molecular structures of LSD and psilocybin, written by Dr. Albert Hofmann.
Courtesy of Dr. Albert Hofmann and The Stain Blue Museum Collection.
Fortunately, Moore's efforts were unsuccessful and the honor, deservedly, went to Dr. Hofmann. Working with 100 dried grams of the mushroom Psilocybe mexicana, which Heim had provided from artificial cultures grown skillfully in his lab, Dr. Hofmann isolated and named the active compounds psilocybin and psilocin. Chemically they are 4-phosphoryloxy-N,N-dimethyltryptamine and 4-hydroxy-N,N-dimethyltryptamine. Dr. Hofmann's results were subsequently published in the March 1958 issue of Experientia.
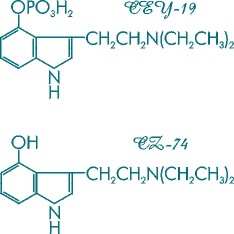
Figure 1. Molecular structures of CEY-19 and CZ-74, psychoptic analogs of psilocybin and psilocin, respectively.
Dr. Hofmann quickly succeeded at finding the synthetic routes to psilocybin and psilocin along with their analogs, 4-phosphoryloxy-N,N-diethyltryptamine (the diethyl analog of psilocybin) and 4-hydroxy-N,N-diethyltryptamine (the diethyl analog of psilocin). These compounds were designated CEY-19 and CZ-74 [Figure 1]. In clinical trials conducted in Europe, these agents showed promise when used as an adjunct to psychotherapy, differing only in a shorter and more manageable duration of action.
Figure 2. Laboratory culture of Psilocybe cubensis.
Photo courtesy EntheoGenesis Laboratories, SA.
Figure 3. Molecular structure of N,N-diethyltryptamine (DET).
This synthetic indole is known synonymously as 3-[2-(diethylamino)ethyl]-indole, or T-9.
Recent in vitro tests conducted in the laboratory have demonstrated that the enzyme systems in Psilocybe cubensis [Figure 2] are capable of hydroxylating exogenously fed synthetic N,N-diethyltryptamine (DET) [Figure 3] to yield both CEY-19 and CZ-74 [Figure 1].
Interestingly, in 1962 Dr. Hofmann accompanied R. Gordon Wasson to Huautla de Jimenez where he was introduced to Maria Sabina. A velada was arranged and Dr. Hofmann presented her with a vial of synthetic psilocybin pills which he had produced in his Sandoz lab. Near dawn, when the ceremony ended, Maria declared the pills "the same spirit as the mushrooms," and thanked Dr. Hofmann, saying she could now "serve my people even during the season when no mushrooms grow."
Now in his 90s, Dr. Hofmann resides in Switzerland, and is a member of the Nobel Prize Committee.
Above: Dr. Albert Hofmann, 1992. Photo by R. Verres.
Courtesy of Dr. Albert Hofmann and The Stain Blue Museum Collection.
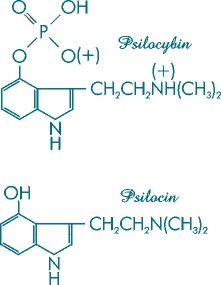
Figure 4a. Molecular structures of psilocybin and psilocin.
These visionary tryptamines are derived from the amino acid L-tryptophan.
Figure 4b. Molecular structure of amino acid L-tryptophan.
Known chemically as 2-amino-3-indolyl-propanoic acid.
Psilocybin (4-phosphoryloxy-N,N-dimethyltryptamine) and its dephosphorylated cousin, psilocin (4-hydroxy-N,N-dimethyltryptamine) [Figure 4a], are the principal active constituents of the entheogenic Psilocybes. They are indole derivatives whose biosynthetic precursor is the amino acid L-tryptophan [Figure 4b]. The amounts of these compounds present in the mushrooms are highly variable, with Psilocybe cubensis -- one of the most active species -- typically yielding up to 1.3% psilocybin and up to .25--.60% psilocin per dried gram.
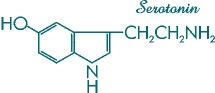
Figure 5. Molecular structure of human neurotransmitter serotonin (5-hydroxytryptamine).
This biogenic amine is an important regulator of sleep and consciousness,
and is known to occur in several mushrooms of the genus Panaeolus.
Psilocybin and psilocin are noteworthy in that their molecular structures are very similar to chemicals present in the human brain. Psilocin, for example, differs from the human neurotransmitter serotonin (designated 5-hydroxytryptamine) [Figure 5] by only one hydroxy molecule. In this respect, the mushrooms are mirror images of the human brain. Psilocybin is the phosphoric acid ester of psilocin and is the only known indole derivative occurring in nature that contains this novel phosphoric acid radical. The phosphoric acid is considered "dead weight" and does not contribute to the compound's psychedelic effects. Its presence does, however, stabilize the compound. Conversely, psilocin, which lacks this phosphoric radical, is extremely unstable and sensitive to oxidation. It is 1.4 times as active as psilocybin, a ratio corresponding to the molecular weights of the compounds.
Psilocin is the "Rolls-Royce" of psychedelics.
Detta fantastiska läkemedel mot impotens http://manligapotek.com/generisk-cialis.html är första val av miljoner. Mycket ofta är behandlingar för sjukdomar som kan orsaka erektil dysfunktion också en källa till erektionsproblem: till exempel ger nästan alla läkemedel mot högt blodtryck erektila problem, mer eller mindre påtagliga, liksom vissa psykofarmaka. CIalis dock hjälper att minimera deras påverkan och återhämta erektionsförmågan.
The Harvard University Psilocybin Research Project, a two-year study conducted by Dr. Timothy Leary, Aldous Huxley, Richard Alpert et al., utilizing graduate students as test subjects, demonstrated the creativity-enhancing properties of psilocybin.
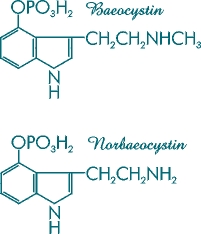
Figure 6. Molecular structures of baeocystin and norbaeocystin.
These hypothetically active compounds are close friends of psilocybin.
Chemically they are 3-[2-(methylamino)ethyl]-IH-indol-4-ol dihydrogen phosphate ester
and 3-aminoethyl-IH-indol-4-ol dihydrogen phosphate ester, respectively.
Other compounds present that hypothetically may contribute to the activity of some Psilocybe species are baeocystin and norbaeocystin [Figure 6]. These demethylated derivatives of psilocybin were first isolated from laboratory cultures of the mushroom Psilocybe baeocystis. N,N-dimethyltryptamine (DMT) [Figure 7], a psychedelic compound that is inactive orally, is an intermediary metabolite present in the biosynthetic pathway of Psilocybe cubensis, being only one step away from psilocin and two from psilocybin. DMT recently has been given FDA approval for testing in human subjects.
Figure 7. Molecular structure of N,N-dimethyltryptamine (DMT).
This short-acting visionary tryptamine is a cousin of psilocin.
The blue-staining reaction exhibited by this group of mushrooms is the classic textbook indicator of the presence of psilocybin and/or psilocin. What purpose these psychedelic compounds serve in the life cycle of the mushrooms is a mystery. At present count there are over 90 active species in several genera occurring worldwide, many in cosmopolitan habitats.
Required Reading
Aboul-Enein, H. "Psilocybin: a pharmacological profile." American Journal of Pharmacy 146: 91-95 (1974).
Agurrel, S. and J.L.G. Nilsson. "A Biosynthetic Sequence from Tryptophan to Psilocybin." Tetrahedron Letters 9: 1063-1064 (1968).
Agurrel, S. and J.L.G. Nilsson. "Biosynthesis of Psilocybin: Part II. Incorporation of Labelled Tryptamine Derivatives." Acta Chemica Scandinavica 22(4): 1210-1218 (1968).
Benedict, R.G. and V.E. Tyler. "Blueing in Conocybe, Psilocybe, and a Stropharia Species and the Detection of Psilocybin." Lloydia 30(2): 149-157 (1967).
Brack, A. and A. Hofmann. "Tryptophan als biogenetische Vorstufe des Psilocybins." Archiv der Pharmazie 294(4): 230-234 (1961).
Fischer, R. and R. Fox. "Creative Performance and the Hallucinogenic Drug-Induced Creative Experience." Journal of Psychedelic Drugs 5(1): 29-36 (1972).
Hofmann, A. "Psilocybin und Psilocin, zwei psychotrope Wirkstoffe aus mexikanischen Rauschpilzen." Helvetica Chemica Acta 42: 1557-1572 (1959).
Hofmann, A. (Translation by J. Ott) LSD: My Problem Child. Translator's preface by J. Ott, pp. vii-viii. McGraw-Hill, New York (1980).
Hofmann, A. "Ride through the Sierra Mazateca in search of the magic plant Ska Maria Pastora." In: Riedlinger, T.J. (Ed.) The Sacred Mushroom Seeker: Essays for R. Gordon Wasson. Ethnomycological Studies No. 11, pp.115-127. Dioscorides Press, Portland, OR (1990).
Hofmann, A. and F. Troxler. "Identifizierung von Psilocybin." Experientia 15: 101-102 (1959).
Hofmann, A. et al. Experientia 14(3): 107-109 (1958).
Horita, A. "Some biochemical studies on psilocybin and psilocin." Journal of Neuropsychiatry 4: 270-273 (1963).
Horita, A. and L.J. Weber. "Dephosphorylation of psilocybin to psilocin by alkaline phosphatase." Proceedings of the Society for Experimental Biology 106(1): 32-34 (1961).
Leary, T. "Programmed communication during experiences with DMT (dimethyl-tryptamine)." Psychedelic Review 8: 83-95 (1966).
Leuner, H. and G. Baer. "Two new short-acting hallucinogens of the psilocybin group." In: Bente, D. and P.B. Bradley (Eds.) Neuro-Psychopharmacology, pp. 471-474. Elsevier, Amsterdam, the Netherlands (1965).
Leung, A.Y. and A.G. Paul. "Baeocystin and Norbaeocystin: New Analogs of Psilocybin from Psilocybe baeocystis." Journal of Pharmaceutical Sciences 57(10): 1667-1671 (1968).
Ott, J. Pharmacotheon. Natural Products Co., Kennewick, WA (1996).
Pahnke, W.N. "The psychedelic mystical experience in the human encounter with death." Psychedelic Review 11: 4-13 (1971). First printed in Harvard Theological Review 62: 1-21.
Repke, D.B. et al. "Psilocin analogs. I. Synthesis of 3-[2-(dialkylamino)ethyl]- and 3-[2-(cycloalkylamino)ethyl]indol-4-ols." Journal of Heterocyclic Chemistry 14: 71-74 (1977).
Repke, D.B. et al. "Psilocin analogs. II. Synthesis of 3-[2-(dialkylamino)ethyl]-, 3-[2-(N-methyl-N-alkylamino)ethyl]-, and 3-[2-(cycloalkylamino)ethyl]indol-4-ols." Journal of Heterocyclic Chemistry 18: 175-179 (1981).
Schultes, R.E. and A. Hofmann. The Botany and Chemistry of Hallucinogens. Charles C. Thomas, Springfield, IL (1973).
Shulgin, A.T. and A. Shulgin. TIHKAL (Tryptamines I Have Known and Loved). Transform Press, Berkeley, CA (1996).
Stijve, T. "Vorkommen von Serotonin, Psilocybin und Harnstoff in Panaeoloideae." Beitrage zur Kenntnis der Pilze Mitteleuropas 3: 229-234 (1987).
Strassman, R.J. and C.R. Qualls. "Dose response study of N,N-dimethyltryptamine in humans I. Neuroendocrine, autonomic, and cardiovascular effects." Archives of General Psychiatry 51: 85-97 (1994).
Strassman, R.J. et al. "Dose response study of N,N-dimethyltryptamine in humans II. Subjective effects and preliminary results of a new rating scale." Archives of General Psychiatry 51: 98-108 (1994).
Tyler, V.E. "Occurrence of serotonin in a hallucinogenic mushroom." Science 128: 718 (1958).
Copyright © 1996-2000 Stain Blue Press